Understanding Certificate Of Analysis (COA) Data For GLP1-T: A Researcher’s Interpretation Guide

If you are procuring synthetic research peptides for laboratory investigation, the Certificate of Analysis (COA) is one of the most critical documents you will ever review. It is the backbone of research integrity, the gatekeeper of reproducible results, and the primary tool for verifying that the GLP1-T you have received meets the specifications required for your study.
For researchers working with GLP1-T, a synthetic dual GIP/GLP-1 receptor agonist peptide used in in vitro and in vivo research models, understanding how to accurately read and interpret a COA is not optional. It is essential.
This guide will walk you through the key data points found on a standard COA for GLP1-T, explain what each metric means in a research context, and equip you with the knowledge to make informed procurement decisions for your laboratory.
Disclaimer: GLP1-T is sold strictly for research purposes only. It is not intended for human consumption, veterinary use, or any therapeutic application. This article is intended solely for professional researchers, academics, and laboratory personnel engaged in lawful scientific inquiry. Nothing in this article constitutes medical advice, a health claim, or a recommendation for personal use of any kind.

What Is a Certificate of Analysis (COA)?
A Certificate of Analysis is a document issued by a manufacturer or an independent third-party analytical laboratory. It provides detailed test results confirming the identity, purity, potency, and quality profile of a specific batch of a chemical or peptide compound.
For research-grade peptides like GLP1-T, the COA serves several vital functions:
- It verifies that the compound matches its stated molecular identity.
- It quantifies the purity of the batch, allowing researchers to calibrate dosing models accordingly.
- It documents the presence or absence of contaminants, degradation products, or residual solvents.
- It provides a traceable record tied to a specific lot or batch number, supporting reproducibility and audit requirements.
Without a valid and transparent COA, no researcher should incorporate a compound into a study protocol. The absence of this documentation introduces unacceptable variables into any experimental design.
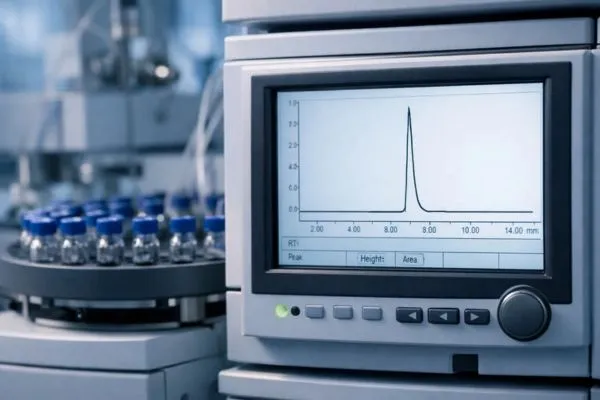
Key Data Points on a GLP1-T COA and How to Interpret Them
The following sections break down the most common analytical metrics you will encounter on a COA for GLP1-T. Each section explains what the test measures, what acceptable values typically look like, and what red flags to watch for.
1. Product Identification
What it includes: Product name, catalog number, Chemical Abstracts Service (CAS) number, molecular formula, molecular weight, and batch/lot number.
Why it matters: Before evaluating any analytical data, confirm that the COA corresponds to the correct compound. GLP1-T has a defined molecular formula and molecular weight. Cross-reference the stated molecular weight and CAS number against published reference databases (such as PubChem or ChemSpider) to confirm identity.
What to verify:
- The molecular formula and weight match known reference values for the GLP1-T peptide sequence.
- The batch/lot number on the COA matches the label on the physical product you received.
- The CAS number, if provided, is consistent with established chemical registry entries.
Any discrepancy at this stage warrants immediate follow-up with the supplier before the compound enters your research pipeline.
2. Peptide Purity (HPLC Analysis)
What it measures: High-Performance Liquid Chromatography (HPLC) is the gold-standard method for assessing peptide purity. It separates the target peptide from any impurities based on differences in chemical interactions with the column’s stationary phase, then quantifies the relative abundance of each component.
Typical acceptable range: Research-grade GLP1-T should generally present a purity of 95% or higher, with many reputable suppliers targeting 98% or above.
How to read the result: The COA will typically report the purity as a percentage. This percentage represents the proportion of the total chromatographic peak area attributable to the target peptide. A result of 98.5% means that 98.5% of the detected material in the sample is the intended GLP1-T peptide, with the remaining 1.5% consisting of related impurities, truncated sequences, or degradation products.
Red flags to watch for:
- Purity below 95% without a clear explanation or intended use case (such as crude peptide for further purification).
- Absence of a chromatogram image or reference to the HPLC method used (C18 column, gradient conditions, detection wavelength).
- Unusually broad or asymmetric peaks, which can indicate co-eluting impurities that inflate the apparent purity.

3. Mass Spectrometry (MS) Confirmation
What it measures: Mass spectrometry confirms the molecular identity of the peptide by measuring its mass-to-charge ratio. Techniques commonly used include Electrospray Ionization Mass Spectrometry (ESI-MS) and Matrix-Assisted Laser Desorption/Ionization Time-of-Flight (MALDI-TOF).
Why it matters: While HPLC tells you how pure the sample is, mass spectrometry tells you whether the predominant compound is actually GLP1-T. It confirms that the peptide has the correct molecular weight, which in turn confirms that the amino acid sequence was synthesized correctly.
How to read the result: The COA should report an observed molecular weight that closely matches the theoretical (calculated) molecular weight of GLP1-T. Minor deviations (typically within +/- 1 Dalton) are acceptable and often attributable to instrument calibration or isotope distribution.
Red flags to watch for:
- A significant deviation between the observed and theoretical molecular weights (greater than 2 Daltons).
- The absence of any mass spectrometry data entirely, which would mean the compound’s identity has not been independently confirmed beyond chromatographic retention time.
- Multiple dominant mass peaks that suggest a mixture of peptides rather than a single, well-defined product.
4. Amino Acid Sequence Verification
What it measures: Some COAs include data from amino acid analysis (AAA) or peptide sequencing techniques that confirm the primary structure of the peptide.
Why it matters: For a complex peptide like GLP1-T, which contains a specific amino acid sequence critical to its receptor binding profile, sequence verification adds an additional layer of identity confirmation beyond molecular weight alone. Two peptides can share a similar molecular weight but differ in sequence, and those differences would alter receptor interaction entirely.
How to read the result: Look for a stated sequence that matches the published reference sequence for GLP1-T. Some COAs may reference sequence coverage percentages from tandem mass spectrometry (MS/MS) fragmentation data.

5. Peptide Content (Net Peptide Weight)
What it measures: Peptide content refers to the actual mass of active peptide in the vial, expressed as a percentage of the total powder weight. The total powder weight includes not only the peptide itself but also counterions (such as trifluoroacetate or acetate salts), residual moisture, and any residual solvents from the lyophilization process.
Why it matters: This is one of the most commonly misunderstood metrics on a COA, and misinterpreting it can directly compromise your experimental accuracy. A vial labeled as containing 10 mg of GLP1-T powder may, depending on the peptide content percentage, contain only 7 to 8 mg of actual peptide.
How to read the result: If the COA states a peptide content of 75%, and the vial contains 10 mg of total lyophilized powder, then the actual peptide present is approximately 7.5 mg. Research protocols that require precise molar concentrations must account for this value when preparing stock solutions.
Action step: Always use the peptide content percentage, not the gross powder weight, when calculating reconstitution volumes and molar concentrations for your experiments.
6. Residual Solvent Analysis
What it measures: This test screens for trace amounts of organic solvents (such as acetonitrile, trifluoroacetic acid (TFA), dimethyl sulfoxide (DMSO), or methanol) that may remain in the lyophilized peptide from the purification and freeze-drying process.
Why it matters: Residual solvents above certain thresholds can interfere with cell-based assays, alter receptor binding kinetics, or introduce cytotoxic effects in in vitro models. The International Council for Harmonisation (ICH) Q3C guidelines provide established limits for residual solvents in pharmaceutical-grade compounds, and these same benchmarks serve as useful reference points for research-grade materials.
How to read the result: The COA should list each tested solvent alongside its detected concentration (usually in parts per million, or ppm) and the applicable limit. Results within ICH guidelines are generally acceptable for most research applications.
7. Endotoxin Testing (LAL Assay)
What it measures: The Limulus Amebocyte Lysate (LAL) assay detects bacterial endotoxins, which are lipopolysaccharides shed from gram-negative bacteria. These endotoxins can provoke strong immune responses in biological systems.
Why it matters: If your research involves cell culture models, primary cell lines, or in vivo animal studies, endotoxin contamination can confound your results by triggering inflammatory pathways unrelated to the compound under investigation. This is especially relevant for studies examining receptor signaling, metabolic pathways, or cellular response mechanisms.
How to read the result: Endotoxin levels are expressed in Endotoxin Units per milligram (EU/mg). For most in vivo research applications, a threshold of less than 5 EU/mg is considered acceptable, though individual study protocols may require stricter limits.
8. Appearance and Solubility
What it includes: Physical description of the lyophilized product (typically a white to off-white powder) and, in some cases, reported solubility in common research solvents.
Why it matters: While this may seem like a minor data point, significant deviations in appearance (such as discoloration, visible particulate matter, or clumping) can indicate degradation, contamination, or improper storage during transit.

How to Use COA Data to Strengthen Your Research Protocols
Understanding the data on a COA is only the first step. The real value lies in applying that information systematically within your laboratory workflow.
Incorporate Peptide Content into All Concentration Calculations
Failing to account for the difference between gross powder weight and net peptide weight is one of the most common sources of dosing error in peptide research. Build this adjustment into your standard operating procedures (SOPs).
Archive Every COA Alongside Your Experimental Records
If your findings are ever audited, peer-reviewed, or challenged, the ability to produce the COA for every batch of every compound used in your study is a powerful safeguard for reproducibility claims.
Compare COAs Across Batches
If you are running a longitudinal study or replicating experiments, request and compare COAs from each batch procurement. Batch-to-batch variability in purity, peptide content, or endotoxin levels can introduce subtle inconsistencies that affect data interpretation.
Request Third-Party Verification When Necessary
If a COA is issued solely by the manufacturer without independent laboratory confirmation, consider submitting a sample to an accredited analytical laboratory for independent testing. This adds cost but provides an additional layer of assurance, particularly for high-stakes or publication-bound studies.

What to Look for in a Reputable GLP1-T Supplier’s COA
Not all COAs are created equal. The difference between a rigorous, transparent COA and a superficial one can mean the difference between defensible research results and months of compromised data. When evaluating a supplier of research-grade GLP1-T, treat the quality of their documentation as a direct reflection of the quality of their manufacturing and quality control processes.
Note: GLP1-T is intended strictly for laboratory research. The evaluation criteria below are designed to help qualified researchers select compounds that meet the analytical standards required for controlled scientific investigation.
Essential Elements Every COA Must Include
A credible COA for research-grade GLP1-T should contain, at a minimum, all of the following elements. The absence of any one of these should prompt further inquiry before integrating the compound into your research workflow.
Unambiguous Product and Batch Identification
The COA must clearly state the product name, molecular formula, molecular weight, CAS number (where available), and a unique batch or lot number. This lot number is the single most important traceability element on the document. It links the analytical data to the specific manufacturing run that produced the material in your hands. Without it, the COA is effectively a general specification sheet rather than a certificate of tested quality.
HPLC Purity Data with Full Method Disclosure
A purity percentage alone is not sufficient. A transparent COA will also disclose the HPLC method parameters used to generate that result. Look for information about the column type (such as C18 reverse-phase), the mobile phase gradient, the detection wavelength (commonly 220 nm for peptides), and the flow rate. This level of detail allows you to assess whether the analytical method was appropriate for the compound and whether the reported purity is truly meaningful. A supplier that reports 99% purity but provides no method details gives you no way to evaluate or reproduce that claim.
Mass Spectrometry Identity Confirmation
The COA should include observed molecular weight data from ESI-MS, MALDI-TOF, or an equivalent technique, alongside the theoretical molecular weight for comparison. This confirms that the compound in the vial is, in fact, GLP1-T and not a truncated sequence, a deletion variant, or an entirely different peptide. Identity confirmation is a non-negotiable element of any credible COA.
Peptide Content (Net Peptide Percentage)
As discussed earlier in this guide, the total powder weight in a vial is not the same as the active peptide weight. A reputable supplier will clearly report the net peptide content as a percentage, enabling you to make accurate concentration calculations. If a COA omits this value, you have no reliable way to prepare precise stock solutions, and every downstream measurement in your study becomes suspect.
Residual Solvent Screening Results
The COA should list the solvents tested, the detected levels in ppm, and the reference limits applied (ideally aligned with ICH Q3C guidelines). This is particularly important for researchers conducting cell-based assays, where even trace amounts of certain solvents can alter cell viability or introduce confounding cytotoxic effects.
Endotoxin Testing (Where Applicable)
For any research involving live biological systems, whether cell culture, primary tissue, or in vivo animal models, endotoxin data is essential. A COA that includes LAL assay results demonstrates that the supplier understands the downstream applications of their product and has taken steps to ensure the material will not introduce inflammatory artifacts into your experiments.
Conclusion
Every data point on a Certificate of Analysis exists to answer a specific question about your GLP1-T compound, its identity, its purity, and its fitness for controlled research. Treat COA review not as a formality but as an active checkpoint in your procurement workflow. Cross-reference molecular weights against published databases, factor net peptide content into every concentration calculation, and flag any missing analytical method details before the compound enters your pipeline. When batch-to-batch variability is a concern, archive and compare COAs systematically to protect the consistency of longitudinal data. Suppliers who provide full HPLC method disclosure, mass spectrometry confirmation, residual solvent screening, and endotoxin results are signaling a commitment to the same analytical rigor your research demands. Build your experimental credibility from the ground up by making COA interpretation a non-negotiable step in every study protocol.
Disclaimer: GLP1-T is sold strictly for research purposes only. It is not intended for human consumption, veterinary use, or any therapeutic application. This article is intended solely for professional researchers, academics, and laboratory personnel engaged in lawful scientific inquiry. Nothing in this article constitutes medical advice, a health claim, or a recommendation for personal use of any kind.
Frequently Asked Questions
What should I do first when I receive a COA for GLP1-T?
Start by verifying product identification. Confirm that the molecular formula, molecular weight, and CAS number on the COA match established reference databases such as PubChem. Then check that the batch or lot number printed on the document matches the label on your physical vial. Do not proceed with any analytical evaluation until these identifiers align, a mismatch at this stage invalidates everything that follows.
How do I use the peptide content percentage to prepare accurate stock solutions?
Multiply the total powder weight in the vial by the peptide content percentage listed on the COA. For example, if you have 10 mg of lyophilized powder and the COA states 75% peptide content, your actual peptide mass is 7.5 mg. Use this adjusted figure, not the gross powder weight, when calculating reconstitution volumes and molar concentrations. Build this conversion into your standard operating procedures to eliminate a common and avoidable source of dosing error.
Why is HPLC purity alone not enough to confirm compound quality?
HPLC quantifies the proportion of the sample attributable to the target peptide, but it does not confirm that the dominant peak is actually GLP1-T. Two different peptides with similar hydrophobic profiles can produce comparable retention times. Mass spectrometry data is required to verify that the observed molecular weight matches the theoretical weight of the GLP1-T sequence. Always look for both HPLC purity and MS identity confirmation on any COA you evaluate.
When is endotoxin testing on a COA critical for my research?
Request and verify endotoxin (LAL assay) data any time your study involves live biological systems, cell culture, primary cell lines, or in vivo animal models. Bacterial endotoxins activate inflammatory signaling pathways that can confound results unrelated to the peptide under investigation. A threshold below 5 EU/mg is generally acceptable for most in vivo applications, but consult your specific protocol requirements, as some assays demand stricter limits.
Should I send GLP1-T samples to an independent lab for third-party testing?
Independent verification is strongly recommended for high-stakes studies, publication-bound research, or any scenario where the COA is issued solely by the manufacturer without external laboratory confirmation. Submit a representative sample to an accredited analytical facility for HPLC purity, mass spectrometry identity, and endotoxin screening. The added cost is modest compared to the risk of building months of experimental work on unverified starting material.
