Assay Design Considerations For Studying Triple-Incretin Analogs Like GLP-3 R

Triple-incretin analogs are synthetic peptide compounds engineered to activate three receptor targets simultaneously: the GLP-1 receptor, the GIP receptor, and the glucagon receptor (GCGR). Unlike single-target or dual-agonist peptides, these polyagonist molecules engage multiple class B G-protein coupled receptor (GPCR) signaling cascades at once, creating layered pharmacological interactions that require specialized experimental frameworks to study effectively.
GLP-3 R is a research-grade triple-incretin analog designed exclusively for in vitro and preclinical laboratory applications. It is not intended for human consumption and is not approved for therapeutic use.
Assay design for triple-incretin analogs like GLP-3 R must account for differential receptor potency, pathway cross-talk between GLP-1R/GIPR/GCGR signaling, and the potential for biased agonism at each target. Researchers studying these compounds typically begin with isolated receptor assays in recombinant cell systems (HEK293 or CHO lines) before progressing to endogenous expression models and ex vivo tissue preparations. Core functional readouts include cAMP accumulation, beta-arrestin recruitment, calcium flux measurement, and CRE-luciferase reporter gene activity.
This guide covers the critical assay design variables that determine data quality and reproducibility when profiling multi-receptor peptide agonists in controlled research settings, including cell platform selection, dose-response curve construction, binding affinity methodology, peptide stability protocols, and experimental control frameworks.
Disclaimer: GLP-3 R is sold strictly for research purposes only and is not intended for human consumption. This article is provided for educational and informational use by professional researchers and academic institutions. Nothing in this article constitutes medical advice, therapeutic guidance, or endorsement of any off-label application. All references to biological activity describe findings observed in controlled laboratory and preclinical settings.
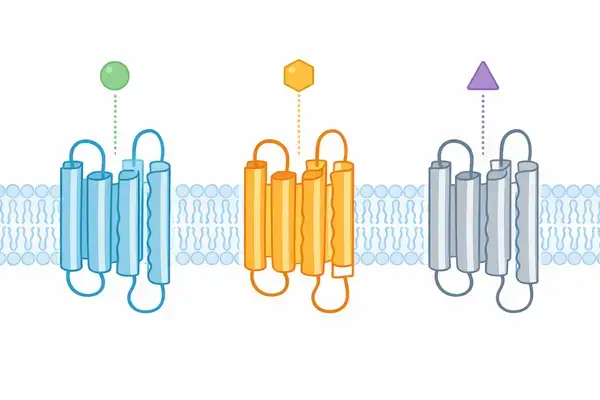
Understanding Multi-Target Receptor Pharmacology in Preclinical Models
Triple-incretin analogs differ fundamentally from single-target or dual-target peptide agonists. Where a mono-agonist activates one receptor subtype and a dual agonist engages two, a triple agonist like GLP-3 R simultaneously interacts with:
- GLP-1 receptors (glucagon-like peptide-1 receptor)
- GIP receptors (glucose-dependent insulinotropic polypeptide receptor)
- Glucagon receptors (GCGR)
Each receptor belongs to the class B G-protein coupled receptor (GPCR) family, yet each initiates distinct downstream signaling cascades. This multi-pathway activation is precisely what makes assay design for triple-incretin analogs more complex than standard single-target peptide studies.
Researchers must account for receptor cross-talk, pathway convergence, and differential potency at each target when designing experiments with compounds like GLP-3 R.
GLP-3 R is intended solely for use in controlled research environments. It is not approved for therapeutic use and must not be administered to humans or animals outside of approved institutional protocols.
Selecting the Right Cell-Based Assay Platform
Recombinant Cell Lines vs. Endogenous Expression Systems
One of the first decisions facing researchers is whether to use engineered cell lines that overexpress a single receptor subtype or cell lines that endogenously express multiple incretin receptors.
Recombinant overexpression systems (such as HEK293 or CHO cells transfected with human GLP-1R, GIPR, or GCGR individually) allow investigators to:
- Measure binding affinity and functional activation at each receptor in isolation
- Generate clean dose-response curves without interference from co-expressed receptors
- Establish selectivity profiles by comparing EC50 values across individual receptor assays
Endogenous expression systems (such as INS-1E beta cell lines or primary pancreatic islet preparations) provide a more physiologically relevant context but introduce confounding variables from simultaneous receptor co-activation and intracellular signal integration.
For initial characterization of a triple-incretin analog like GLP-3 R, most experienced investigators begin with isolated receptor assays in recombinant systems before advancing to more complex endogenous models.
Recommended Functional Readouts
The following functional assay readouts are commonly employed when profiling multi-receptor incretin agonists in research settings:
- cAMP accumulation assays (HTRF, LANCE, or AlphaScreen formats) to measure Gs-coupled receptor activation
- Beta-arrestin recruitment assays (PathHunter or TANGO systems) to evaluate biased agonism
- Calcium flux assays (FLIPR or plate-based fluorometric readers) to detect Gq-mediated signaling, particularly relevant for GCGR studies
- Reporter gene assays (CRE-luciferase constructs) to assess transcriptional activation downstream of cAMP signaling
Each of these platforms offers different sensitivity thresholds, dynamic ranges, and throughput capabilities. Selecting the appropriate readout depends on the specific research question and the receptor target under investigation.
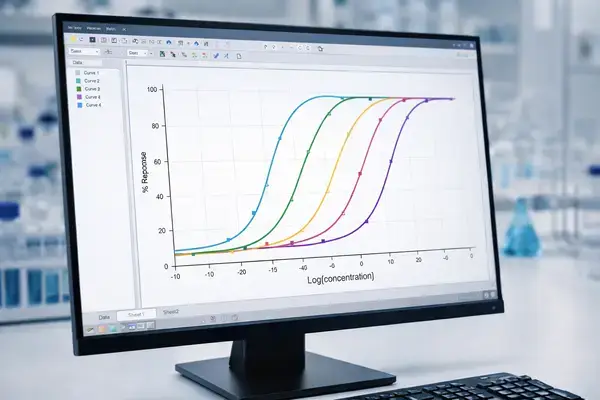
Dose-Response Curve Construction and EC50 Determination
Generating reliable concentration-response data is essential for characterizing the pharmacological profile of any multi-target peptide agonist. When working with GLP-3 R or similar triple-incretin analogs, researchers should consider the following variables:
Concentration Range Selection
A broad concentration range (typically 10⁻¹² M to 10⁻⁵ M) with half-log or third-log dilution steps provides the resolution needed to capture full sigmoidal curves across all three receptor targets. Triple agonists may exhibit different potency at each receptor, meaning the curve inflection point will shift depending on which target is being measured.
Incubation Time Optimization
Signal kinetics vary across GLP-1R, GIPR, and GCGR. cAMP accumulation may peak at different time points for each receptor subtype. Researchers should perform time-course experiments during assay development to identify the optimal incubation window that captures maximal signal-to-noise for each receptor independently.
Curve Fitting and Statistical Analysis
Four-parameter logistic (4PL) regression models remain the standard for fitting sigmoidal dose-response data. When comparing potency across receptors, researchers should report:
- EC50 values with 95% confidence intervals
- Hill slope coefficients (which may differ across receptor subtypes)
- Emax values relative to a validated reference agonist for each receptor
Using a reference standard (such as native GLP-1(7-36)NH2 for GLP-1R, native GIP(1-42) for GIPR, and native glucagon for GCGR) alongside GLP-3 R in every experiment enables normalization and cross-study comparison.

Binding Affinity Studies: Radioligand and Fluorescence-Based Approaches
Functional assays measure downstream signaling, but receptor binding studies provide direct information about ligand-receptor interactions at the molecular level.
Radioligand Competition Binding
Saturation binding experiments using radiolabeled tracers (such as ¹²⁵I-labeled reference ligands) can determine the equilibrium dissociation constant (Kd) of GLP-3 R at each target receptor. Competition binding assays, where increasing concentrations of unlabeled GLP-3 R displace a fixed concentration of radiotracer, yield Ki values that quantify binding affinity independent of downstream signaling amplification.
Fluorescence Polarization and TR-FRET Alternatives
For laboratories without radioligand capabilities, fluorescence-based binding assays offer a viable alternative. Time-resolved FRET (TR-FRET) and fluorescence polarization (FP) assays using labeled peptide tracers can provide Kd and Ki estimates with comparable accuracy, while avoiding the regulatory and safety considerations associated with radioisotope handling.
Addressing Biased Agonism in Triple-Incretin Research
An increasingly important consideration in incretin receptor research is the concept of biased agonism, where a ligand preferentially activates one signaling pathway over another at the same receptor.
Triple-incretin analogs like GLP-3 R may exhibit distinct bias profiles at each of their three target receptors. For example, a compound might preferentially activate Gs-mediated cAMP signaling over beta-arrestin recruitment at GLP-1R, while showing the opposite bias pattern at GCGR.
To assess biased agonism, researchers should:
- Measure at least two signaling endpoints at each receptor (typically cAMP and beta-arrestin)
- Calculate bias factors using the operational model of agonism (Black and Leff framework)
- Compare bias factors against a balanced reference agonist for each receptor subtype
Understanding signaling bias has significant implications for interpreting the preclinical pharmacology of multi-target peptide agonists and may help explain differential downstream effects observed in tissue-level or whole-organism research models.

Peptide Stability and Handling Considerations for Assay Reproducibility
Research-grade peptide analogs require careful handling to maintain structural integrity and biological activity throughout experimental workflows.
Reconstitution and Storage
- Reconstitute lyophilized GLP-3 R in sterile bacteriostatic water or an appropriate buffer (such as PBS at pH 7.4) according to the product’s certificate of analysis
- Prepare single-use aliquots to avoid repeated freeze-thaw cycles, which can promote peptide aggregation and loss of activity
- Store reconstituted aliquots at -20 degrees C or below for long-term retention; use 2 to 8 degrees C for short-term working stocks (less than 7 days)
Adsorption and Carrier Protein Considerations
Hydrophobic or amphipathic peptides can adsorb to plastic labware surfaces, leading to unpredictable concentration losses. Researchers should consider:
- Using low-binding microcentrifuge tubes and plate formats
- Adding carrier proteins (such as 0.1% BSA) to assay buffers to minimize nonspecific surface adsorption
- Validating peptide recovery at each handling step through analytical methods like reverse-phase HPLC or mass spectrometry
These precautions are especially important at low nanomolar and picomolar concentrations, where surface losses represent a larger fraction of total peptide in solution.
Experimental Controls and Data Validation
Robust experimental controls are the backbone of credible preclinical peptide research. When working with GLP-3 R, the following controls should be integrated into every assay:
- Positive controls: Known reference agonists for each receptor (native GLP-1, GIP, and glucagon peptides at validated concentrations)
- Negative controls: Vehicle-treated wells (buffer without peptide) and, where possible, cells expressing a non-target receptor to confirm specificity
- Antagonist controls: Selective receptor antagonists (such as exendin(9-39) for GLP-1R, or des-His-glucagon for GCGR) can confirm that observed signals are receptor-mediated
- Assay performance metrics: Calculate Z-prime (Z’) values for each plate to verify adequate separation between positive and negative controls (Z’ greater than or equal to 0.5 is generally considered acceptable for screening assays)
Advancing from In Vitro to Ex Vivo and Tissue-Level Studies
Once receptor-level pharmacology is established through cell-based assays, researchers often seek to evaluate peptide activity in more complex biological contexts.
Isolated Tissue Preparations
Perfused organ systems (such as isolated pancreatic islet preparations or perfused liver models) allow investigators to study how triple-receptor activation by GLP-3 R translates into integrated tissue-level responses. These ex vivo systems preserve native receptor density, co-expression patterns, and intercellular signaling networks that are absent in recombinant cell lines.
Primary Cell Cultures
Freshly isolated primary cells (including human or rodent pancreatic beta cells, hepatocytes, or adipocytes) provide another layer of physiological relevance. However, primary cell variability between donor preparations necessitates larger sample sizes and careful normalization strategies.
All tissue-level and primary cell studies involving GLP-3 R should be conducted under appropriate institutional oversight and in accordance with applicable biosafety and ethical review protocols.

Building a Rigorous Assay Framework for Triple-Incretin Analog Research
Studying triple-incretin analogs like GLP-3 R requires a methodical, multi-layered approach to assay design. Researchers who invest time in optimizing their cell-based platforms, validating dose-response parameters, integrating appropriate controls, and addressing peptide handling variables will generate data that is both reproducible and meaningful within the broader context of incretin receptor pharmacology.
The complexity of multi-receptor agonism demands that investigators move beyond a single-assay mindset and instead build comprehensive pharmacological profiles that capture activity, selectivity, potency, and signaling bias across all three receptor targets.
GLP-3 R provides a valuable research tool for investigators studying the intersection of GLP-1, GIP, and glucagon receptor biology in controlled laboratory settings.
Conclusion
Triple-incretin analog research sits at a critical inflection point where multi-receptor pharmacology demands more from experimental design than traditional single-target studies ever required. Investigators who build their assay frameworks around isolated receptor profiling, validated dose-response protocols, and layered functional readouts will position their laboratories to generate data that withstands peer review and advances the broader understanding of polyagonist peptide biology. The variables outlined in this guide (cell platform selection, binding affinity methodology, biased agonism assessment, peptide stability controls, and statistical validation) are not optional refinements. They are baseline requirements for any research team working with compounds like GLP-3 R. Start with recombinant systems, validate with endogenous models, and progress methodically toward ex vivo preparations. That progression, executed with disciplined controls at every stage, is what separates publishable incretin receptor data from noise.
FAQs
What is GLP-3 R and what receptor targets does it engage?
GLP-3 R is a synthetic triple-incretin analog that simultaneously activates the GLP-1 receptor, the GIP receptor, and the glucagon receptor (GCGR), all of which belong to the class B GPCR family. This polyagonist activity profile makes it a valuable tool for researchers investigating multi-receptor signaling interactions in preclinical laboratory models. GLP-3 R is sold strictly for research purposes only and is not intended for human consumption or therapeutic use.
What cell-based assay platforms are recommended for studying triple-incretin analogs like GLP-3 R?
Researchers typically begin with recombinant overexpression systems such as HEK293 or CHO cells transfected with individual receptor subtypes (GLP-1R, GIPR, or GCGR) to isolate activity at each target independently. Once single-receptor profiles are established, investigators can advance to endogenous expression systems like INS-1E beta cell lines or primary pancreatic islet preparations for more physiologically relevant data. Core functional readouts include cAMP accumulation assays, beta-arrestin recruitment assays, calcium flux measurement, and CRE-luciferase reporter gene activity.
How should researchers handle and store GLP-3 R to maintain peptide stability during experiments?
Reconstitute lyophilized GLP-3 R in sterile bacteriostatic water or PBS buffer at pH 7.4, then divide the solution into single-use aliquots to prevent degradation from repeated freeze-thaw cycles. Store aliquots at negative 20 degrees Celsius or below for long-term retention, and keep short-term working stocks at 2 to 8 degrees Celsius for no longer than seven days. Use low-binding labware and add 0.1% BSA to assay buffers to minimize peptide adsorption to plastic surfaces, especially at low nanomolar concentrations.
What experimental controls should be included when running assays with GLP-3 R?
Every assay should include positive controls (native GLP-1, GIP, and glucagon peptides at validated concentrations), negative controls (vehicle-treated wells and non-target receptor expressing cells), and antagonist controls (such as exendin(9-39) for GLP-1R) to confirm receptor-mediated signaling. Calculate Z-prime values for each plate to verify that signal separation between positive and negative controls meets the accepted threshold of 0.5 or higher. These layered controls allow researchers to distinguish true receptor activation from assay artifacts and ensure that results are reproducible across independent experiments.
Why is biased agonism relevant when studying triple-incretin analogs in preclinical research?
Biased agonism occurs when a peptide ligand preferentially activates one intracellular signaling pathway over another at the same receptor, and triple-incretin analogs like GLP-3 R may display different bias profiles at each of their three targets. Researchers can quantify this by measuring at least two signaling endpoints per receptor (typically cAMP accumulation and beta-arrestin recruitment) and calculating bias factors using the Black and Leff operational model. Characterizing signaling bias is essential for interpreting how multi-receptor activation translates into differential downstream effects in tissue-level and whole-organism research models.
